9210 Drug-Eluting Stent Test Instrument0 pages
ElectroForce® 9210 Drug-Eluting
tttttStent Test Instrument
Accelerated Pulsatile Distension
tttfor Coated Stent Durability
Ten-year FDA accelerated bench testing of coated stents can be performed in weeks
with the ElectroForce® 9210-12 drug-eluting stent test instrument. This application
of ElectroForce high bandwidth linear motors provides the capability to test more
stents faster. The modular design allows the stent developer to acquire additional
test capabilities as they are needed. The test modules consist of:
• The ElectroForce 9210-12 pulsatile fatigue instrument
• The automated particle capture module
• Interface to real-time particle counters.
The base ElectroForce drug-eluting stent/graft test instrument provides several
test capabilities to the stent developer, including:
• Accelerated 10 to 15 year (400-600 million cycles) fatigue
durability testing at relevant physiological distensions
• Accelerated fatigue to fracture testing at greater than
physiological distensions
• Demonstrated performance bandwidth to perform physiological or
accelerated (from 1 Hz to >100 Hz*) durability testing of coated stents.
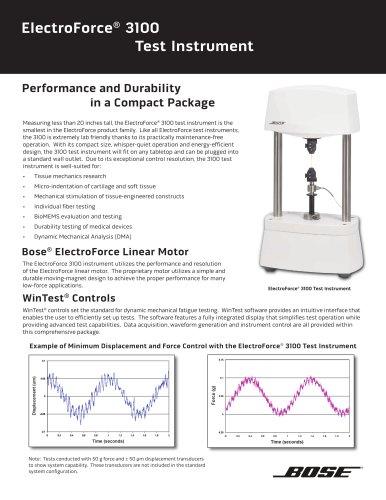
ElectroForce®
Model 9210-12
Drug-Eluting Stent
Pulsatile Fatigue Instrument
Features and Benefits
•
Accelerated closed-loop servo-control of pulsatile distension
•
Up to 12 mock arteries per test
- This leaves as many as 10 tubes available for testing when using
the recommended ‘blank’ tube and optional ‘bare metal stent’ tube
•
2.0 mm to 14.0 mm mean device diameter capacity*
•
Adjustable tube length for optimal testing of longer or shorter stents
•
Programmable control and system monitoring using WinTest® Controls.
This provides the user with real-time computer control and monitoring of:
- Distension control
- Pressure control
- Flow rates
Laser micrometer for measurement and
control of pulsatile distension
- Filter condition and automated filter changes
Direct diametric distension measurement using the integrated
laser micrometer
•
*Maximum distension levels and test frequencies dependent
on tube diameter, wall thickness, and length.
•
Meets or exceeds international standards such as ISO 25539 and ASTM
F2477 as well as related FDA guidance documents.

 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید