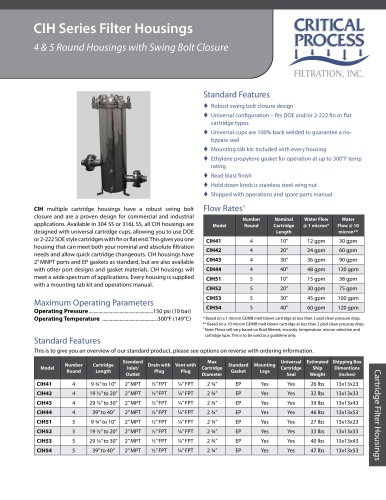
Biopharm PES Cartridge Filters0 pages
BPS Cartridge Filters
PES Membrane
Applications
Buffers and Feedstocks
SVPs
WFI Water
LVPs
Solvents
Vaccines
Length
5 to 40 in. (12.7 to 101.6 cm) nominal
Outside Diameter
2.75 in. (7.0 cm) nominal
Filtration Area
7.0 ft2 (0.65 m2) per 10 in. length
Integrity Test Specifications
10-inch cartridge, water wetted membrane
Pore Size
0.03 μm
Biopharmaceutical grade PS filters are hydrophilic and
manufactured with asymmetric polyethersulfone (PES)
membrane. PES membrane exhibits excellent flow rates with high
throughput. BPS cartridge filters are used for critical bioburden
reduction applications in the biopharmaceutical industry. Specific
applications for BPS filters are bacteria removal in buffers, USP
Water for Injection (WFI), SVPs, LVPs and other biopharmaceutical
products. The filter elements are 100% integrity tested during
production.
Polyethersulfone is particularly suited for the filtration of
products that contain elements that adsorb to the media, such as
preservatives and proteins. The lower binding characteristics of
polyethersulfone make it a good choice for filtration of valuable
protein solutions such as vaccines and biologicals.
Construction Materials
Filtration Media
Polyethersulfone (PES) Membrane
Media Support
Polypropylene
End Caps
Polypropylene
Center Core
Polypropylene
Outer Support Cage
Polypropylene
Sealing Method
Thermal Bonding
O-rings
Buna, Viton® (or FKM), EP, Silicone,
FEP Encapsulated Silicone, FEP
Encapsulated Viton (or FKM)
Air Diffusion Rate
< 30 cc/min at 60 psig (4.1 barg)
0.10 μm
< 30 cc/min at 48 psig (3.3 barg)
0.22 μm
< 30 cc/min at 35 psig (2.4 barg)
0.45 μm
< 30 cc/min at 20 psig (1.4 barg)
0.65 μm
< 30 cc/min at 15 psig (1.0 barg)
0.80 μm
< 30 cc/min at 12 psig (0.8 barg)
1.0 μm
< 30 cc/min at 8 psig (552 mbarg)
1.2 μm
< 30 cc/min at 7 psig (483 mbarg)
Maximum Operating Parameters
Differential Pressure
•t Forward
50 psid (3.4 bard) at 20 °C (68 °F)
•t Reverse
40 psid (2.7 bard) at 20 °C (68 °F)
Operating Temperature
Recommended
Changeout Pressure
82 °C (180 °F) at 10 psid
(0.69 bard) in water
35 psid (2.4 bard)
Sanitization/Sterilization
Filtered Hot Water
90 °C (194 °F), 30 minutes, multiple
cycles, max 3 psid forward flow
Autoclave
121 °C (250 °F), 30 min, multiple cycles
In-line Steam
135 °C (275 °F), 30 min, multiple cycles
For all elevated temperature procedures above, a stainless steel
support ring is required.
Chemical Sanitization
Performed using industry standard concentrations of hydrogen
peroxide, paracetic acid, sodium hypochlorite, and other
selected chemicals.
Biopharmaceutical Grade
Dimensions
"
 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید