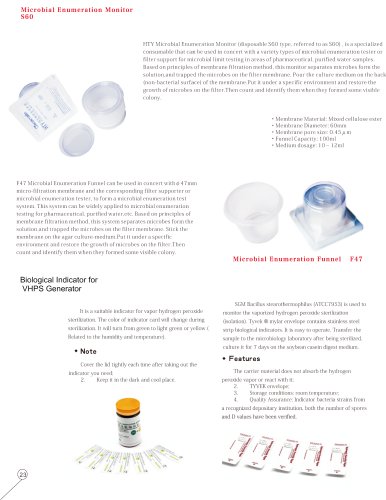
Tailin Sterility Test Isolator System; Sterile Isolator for Production/ Restricted Access Barrier Systems0 pages
Technical parameters
New Sterility Test Isolator System
STERILE ISOLATOR FOR PRODUCTION
Features
☞Soft cabin structure made of transparent PVC membrane, the top overall with closed structural design, integrated control
unit, intake and exhaust systems, air filtration units, etc., more elegant appearance, easy to do cleaning and maintenance;
☞Operation cabinet is designed with standard operating panel of 8 gloves, four primary ones and four secondary ones; the
sterile transfer passageway is designed with 4 standard operating panels, and it optimized the operation of ergonomics
requirements, no operating blind zones;
☞Oversleeves are made from Hypalon ® material, which further improved the isolator’s impermeability, chemical
compatibility and mechanical abrasion resistance;
☞The system supports two Pharmacopoeia sterility test methods: membrane filtration method and the direct inoculation
method. The sterility test pump unit is directly integrated into the instrument operating platform;
☞Operating cabin, transfer passageway can be used together or alone;
☞Cabin intake and exhaust systems adopt H14 grade HEPA and high-performance centrifugal fans imported from Germany.
The cabin airflow pattern is turbulence, which is able to maintain a positive pressure continuously;
☞The system is installed with the function as efficiency filter pressure monitoring, and can display HEPA ventilation status in
real-time ;
☞The system reserves an interface and a valve for connecting the Vapor Hydrogen Peroxide Generator (VHPS ®); the internal
part is sterilized by hydrogen peroxide vapour, the effectiveness of killing spores in the cabin air and the exposed surface
achieves lg6 ;
☞It can communicate with HTY-V100 Vapor Hydrogen Peroxide Generator online in real time , can control the generator on
the isolator control panel;
☞Deliver goods through various ways, for instance, docking to the transfer bag of being able to be steam sterilized , or to the
transfer passageway with RTP system , etc. ;
☞With larger diameter material door design, more convenient for passing materials and cleaning the cabin ;
☞Can choose to install the sterile waste delivery channel, transfer all types of testing waste out timely , which help to save
operating rooms;
☞The system adopts Siemens PLC control system and touch-sensitive color display module, with optimized interface design,
the system operates automatically, and with decompression alarm function;
☞With multifunction interfaces, such as 25mm standard interface, public media (electricity, gas) and verification / testing
interface;
☞Verification: abide to and comply with USP 1208 validation requirements;
☞With multilevel user login program, electronic data storage records and exports functions, the software design meets FDA
21CFR Part 11 regulations.
17
PRODUCT CATALOGUE
Hangzhou Tailin Bioengineering Equipments Co., Ltd.
has been committed to the development of isolation
technology and the applied research of pharmaceutical
industry for a long time. We can provide comprehensive
technology solutions with a variety of isolators, ranging from
the QC department’s sterility test technology, biosafety
containment, the isolator technology for production (sterile
packing, weighing, ingredients, crushing, sampling, etc.) to
RABS. The latest sterile isolators for QC and R & D laboratory
detection, is almost suitable for all sterile testing of sterile
preparations and sterile bulk drugs (API),which based on the
pharmacopoeia statutory methods (membrane filtration
method and the direct inoculation method).
☞Power supply: AC 220V/50Hz
☞Power: 1500W
☞Touch screen: Siemens 7.5 inches color touch screen
☞Cabin pressure control range:-80Pa ~ 80Pa
☞Humidity Resolution: 0.1%
☞Temperature Resolution: 0.1 ℃
☞Pressure Resolution: 0.1Pa
☞Plenum chamber micro-differential pressure gauge resolution: 10Pa
☞PC Connection distance: ≤ 100m
☞Built-in sterility test pump maximum flow : ≥ 300ml/min
☞purification level within cabin: A grade
☞Impermeability: leakage rate per hour ≤ 0.5%
☞Material door diameter: 500mm
☞Transfer gate diameter: 280mm
☞Exhaust pipe diameter: 50mm
☞Basic dimensions: Experiment Module: (L * W * H)1806mm * 1048mm *2000mm; Passing Cabin: (L* W * H)1306mm *
1048mm * 2000mm
☞Weight: Experiment Module: about 270Kg ; Pass cabin: about 200Kg
/RESRICTED ACCESS BARRIER SYSTEMS
Isolation system is specially designed based on product's
characteristic and requirements in the production process .
Application areas
☞Sterile-packing: mainly used in filling
liquid formulations, and solid powder
packing
☞Aseptic ingredients;
☞Aseptic sampling
☞Aseptic weigh
☞Grinding, mixing, and grinding;
☞Provide an aseptic conditions for aseptic
and toxic products;
☞Provide an aseptic conditions for nonaseptic and toxic products.
Technical support
☞Comply with the 2010 version of GMP(Good Manufacturing Practice);
☞Special attention should be paid to when goods are passed in and out of the
isolator;
☞Isolator environment depends on its design and application, and it is
required at least shall be class D clean area;
☞Hangzhou Tailin Bioengineering Equipments Co., Ltd provides a full range of
custom services, including the design, manufacture, installation,
commissioning and maintenance of isolation systems for production. We will
cooperate closely with you and provide with solutions in line with your
expectations and specific requirements.
18
"
 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید