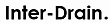"
00
MEASUREMENT AND TESTING
Analysis of Impurities in Polymer-grade
Ethylene, Propylene and 1,3-Butadiene
by Surinder (Sandy) Thind
Current Detection Techniques –
A Case Study with Arsine:
Abstract:
Producers of high purity monomers need to measure, not
only, more and more impurities in their high purity product,
but also, they need to measure these impurities at lower and
lower detection limits. This paper describes the latest
technique available to perform these analyses at low ppb
levels with high levels of accuracy and speed. Both laboratory
and on-line detection is possible.
The Critical Nature of Impurity Analysis:
The production of poly-ethylene and poly-propylene has
become a very competitive business. Producers utilize more
selective and more sensitive polymerization catalysts all the
time. These catalysts are very expensive, and frequent
replacements may lead to a loss. During catalyst replacement,
the plant is shut down, which further adds to losses. In order
to avoid frequent shutdowns due to catalyst poisoning, these
companies insist that monomer producers meet tight
specifications regarding such poisonous impurities. Failure to
comply can result in lawsuits and loss of business. For this
reason, impurity analysis has achieved critical importance.
Take as a case study the detection of one impurity: arsine in
olefins. Even at low ppb levels, arsine can adversely affect
certain polymerization catalyst properties and also lead to
polymer contamination.
Detection of arsine in ethylene, propylene, or 1,3butadiene is very difficult at the desired 5 ppb level. Normally,
a complicated GC column system is used to separate arsine
from propylene. After this separation of low ppb levels of the
impurity from almost 99.9% propylene, expensive systems
such as GC-AED, are used to these ppb levels of arsine. Since
GC-AED will also respond to ppb or ppm levels of other
impurities, such as hydrogen cyanide, nitric oxide, ammonia, or
hydrogen chloride. This being the case, with many other
impurities present at low ppb levels, it is easy to misidentify
the arsine peak. Thus, the individual working with GC-AED
must be highly skilled. Even the most experienced chemists
experience difficulty in positively identifying and accurately
quantifying low (1-10 ppb) levels of this impurity. As a result,
most companies have stopped using this technique for arsine
detection and have gone back to the ‘old’ wet technique
method, which requires hours of bubbling followed by
detection via atomic absorption.
The manual laboratory technique and the GC-AED
technique for ppb-level arsine detection are only available for
laboratory testing applications to date. Further, both of these
techniques are not only impractical for on-line applications,
but they are also very expensive to install and maintain. It is
clear that, in today’s monomer-production facilities, nonspecialist technicians must be able to quickly, efficiently, and
accurately perform arsine analysis without GC separations
both either in the lab or on-line.
It is at this point that the time-proven analytical technique
of Dry Colorimetry draws new attention. The application of
the dry colorimetric technique to the measurement of trace
arsine in the hydrocarbon streams has been independently
demonstrated by several petroleum/petrochemical companies.
Figure 1 indicates potential monitoring points in an
olefin/polyolefin plant for trace arsine measurements. With
prompt, reliable on-line results, the process control engineer
has the opportunity to develop strategies to minimize the
effect of a periodic excursion in arsine levels.
In one field test, the concentration of arsine was monitored
before and after an arsine scrubber.Wet chemical analysis was
used to obtain the concentration of arsine, 25 ppb and 0 ppb,
respectively. The results obtained using Dry Colorimetry
technology corresponded to the wet chemical results within
experimental error.
A New Look at Dry Colorimetry:
INTRODUCTION
Classical colorimetry utilizes an impinger to collect gas in a
liquid medium. Chemical reagents are then added to the
medium to cause it to change color in proportion to the
concentration of gas present. The resulting color change is
measured by a laboratory spectrophometer and compared to
known standards.
Ultra-sensitive "tape" detectors, are also colorimetric
based, but these are dry reaction substrates that serve as gas
collecting and analyzing media. Individually formulated for a
specific gas or family of gases, each detection tape is a nontoxic, proprietary chemical reagent system.When exposed to
a target gas, the tape will change color in proportion to the
amount of gas: the higher the target gas concentration, the
darker the stain that will appear.
The change in color, or stain, on the tape is read by a
photo-optical system in the analysis instrument, and the
intensity of this stain is then compared to a standard response
curve preprogrammed into the instrument’s data system.
Non-Hydrocarbon Impurities
That Require Monitoring:
Just one of the impurities listed in Table 1 has the power to
adversely affect many polymerization catalysts, yet more than
one of these may be present during the process. For this
reason, monomer producers must be able to accurately
detect and measure one or more of the specified impurities
at very low levels.
Impurity
Typical Specification
Arsine
Less than 20 ppb
Phosphine
Less than 20 ppb
Ammonia
Less than 100 ppb
Hydrogen Sulfide
Less than 20 ppb
Carbonyl Sulfide
Less than 20 ppb
Nitrogen Dioxide
Less than 50 ppb
HCN
Less than 100 ppb
HCL, HF
Less than 200 ppb
Phosgene
Less than 50 ppb
Sulfur Dioxide
Less than 50 ppb
Chlorine
Less than 30 ppb
Table 1: Typical Impurities That Require Monitoring in
Monomer Production
PIN June/July 2003
Figure 1: Some Potential Monitoring Points for Arsine in an Olefin /Polyolefin Plant
"

 عضویت
عضویت  ورود اعضا
ورود اعضا راهنمای خرید
راهنمای خرید